In another life, I taught high school biology and had a lot of fun doing it. I had my students do the Cell City when we worked with organelles in the cell, and once we got to the genetics unit, we did something fun called Dragon Genetics. In this activity, students would pair up (one was the mommy dragon, the other the daddy dragon) and throw “chromosome” sticks to see what traits they would “pass on” to their theoretical dragon baby. The activity is quite simple once students understood basic Mendelian genetics (and some of the non-Mendelian patterns as well), and even my son was able to draw his own dragon baby when I had him be my guinea pig while he was still in elementary school. (Figure 1) There were some amazingly creative dragons adorning my classroom, and I hope you can share the Dragon Genetics activity with any teacher friends as we discuss non-Mendelian traits and disease here. As we celebrate the beautifully-designed experiments by Gregor Mendel that led to the modern study of genetics and genomics, we might also be reminded that patterns of inheritance, like many things in life, are far from binary.

Figure 1: My son’s dragon that he drew when doing the Dragon Genetics activity when he was 10 years old.
(Editor’s note: This article originally published on July 20, 2022, the 200th birthday of Gregor Mendel, the father of modern genetics, whose simple yet elegant experiments led to so much more than he could have imagined.)
If Only It Were Simpler: Mendelian Genetics As the Exception?
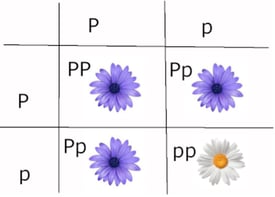 Most people have their first experience with genetics while setting up Punnett squares, with the assumption that there is dominance, only two alleles, and that the dominant allele is fully penetrant, which means if the organism has that allele, the trait will be expressed. (Figure 2) This relatively simple exercise was inspired by the first large-scale genetics experiments performed by Gregor Mendel over 150 years ago, from which he derived the laws of Mendelian inheritance.
Most people have their first experience with genetics while setting up Punnett squares, with the assumption that there is dominance, only two alleles, and that the dominant allele is fully penetrant, which means if the organism has that allele, the trait will be expressed. (Figure 2) This relatively simple exercise was inspired by the first large-scale genetics experiments performed by Gregor Mendel over 150 years ago, from which he derived the laws of Mendelian inheritance.
Figure 2: An example of a Punnett square with full dominance, where the dominant allele will completely mask the recessive allele in the heterozygote.
For those who understand experimental bias, Mendel purposefully chose pure-breeding plants with fully penetrant traits, so he did not have to deal with the diverse patterns of inheritance due to sex linkage, polygenic traits, codominance, and other “non-Mendelian” mechanisms. However, Mendel’s studies using thousands of plants provided the groundwork for our understanding of how genes are passed down through generations and helped us understand how meiosis creates diversity.
As we know in biology, nothing is ever that simple. Most traits have multiple alleles, including the famous ABO blood group antigens. Many human traits are polygenic, or controlled by multiple genes, including those governing our appearance, such as eye color and body shape. Other traits are governed by epistasis, in which two or more genes can affect the expression of that trait. One disease that is influenced by epistatic factors is sickle cell anemia. 1 While often well studied in other organisms, epistasis is not as well understood in humans. 2
 Most traits are determined by the gene sequences stored in our genomic DNA, but there are some exceptions, chiefly the separate DNA found in mitochondria. (Figure 3) Our mitochondria are donated from the mother, which is why ancestry service sites often provide mitochondrial DNA (mtDNA) as an option to trace maternal lineages. While the amount of mtDNA and the size of the mitochondrial genome is dwarved by the main host genome, mutations in mtDNA can contribute to clinical syndromes, some of which can cause blindness and deafness as well as susceptibility to certain diseases. There are also epigenetic factors, including parental imprinting, that could determine which copy of a gene is active while the other is silenced in a non-random manner. The complex interplay between unique genes and environmental factors hinders the diagnosis and treatment of human genetic disorders. 2
Most traits are determined by the gene sequences stored in our genomic DNA, but there are some exceptions, chiefly the separate DNA found in mitochondria. (Figure 3) Our mitochondria are donated from the mother, which is why ancestry service sites often provide mitochondrial DNA (mtDNA) as an option to trace maternal lineages. While the amount of mtDNA and the size of the mitochondrial genome is dwarved by the main host genome, mutations in mtDNA can contribute to clinical syndromes, some of which can cause blindness and deafness as well as susceptibility to certain diseases. There are also epigenetic factors, including parental imprinting, that could determine which copy of a gene is active while the other is silenced in a non-random manner. The complex interplay between unique genes and environmental factors hinders the diagnosis and treatment of human genetic disorders. 2
Figure 3: Mitochondria: the powerhouse of the cell thanks mostly to mom.
Diseases With Non-Mendelian Inheritance
Honestly, the list could encompass most diseases, which are rarely caused by just one mutation or overexpressed gene product. As an example, in the Japanese population, there is a hereditary retinal degenerative disorder in which the retinitis pigmentosa gene RP1, which is frequently mutated in this visual impairment diseases. A screen of patients and carriers of these mutations showed that while most variants followed normal Mendelian patterns of inheritance, a particular variant termed “m3” was passed along in a non-Mendelian manner. 3 Additionally, m3 could interact with the other Mendelian variants (and vice versa) in a non-Mendelian pattern to modulate the disease phenotype.
 It is logical that the nervous system, one of the most complex organ systems ever evolved, would also have diseases that derive from non-Mendelian inheritance patterns due to the interactions of thousands of genes among the billions of neurons. One such type of disease is collectively termed Charcot-Marie-Tooth neuropathy (CMT), which causes damage mostly in the peripheral nervous system and results in smaller, weaker muscles and difficulty in locomotion. While genes associated with CMT have been identified and characterized, a Mendelian hypothesis is not always supported as the genetic confirmation of a positive diagnosis is far from 100%. 4 The same is true of other neurological disorders, including pediatric-onset disorders, ALS, and Parkinson’s disease. 4-6
It is logical that the nervous system, one of the most complex organ systems ever evolved, would also have diseases that derive from non-Mendelian inheritance patterns due to the interactions of thousands of genes among the billions of neurons. One such type of disease is collectively termed Charcot-Marie-Tooth neuropathy (CMT), which causes damage mostly in the peripheral nervous system and results in smaller, weaker muscles and difficulty in locomotion. While genes associated with CMT have been identified and characterized, a Mendelian hypothesis is not always supported as the genetic confirmation of a positive diagnosis is far from 100%. 4 The same is true of other neurological disorders, including pediatric-onset disorders, ALS, and Parkinson’s disease. 4-6
Beyond Mendel: The Exploration of Big Picture Genetics
While next-generation sequencing and other whole genome technologies have facilitated the identification of causative factors in these disorders, and diagnostic yields are extremely low, which suggests the need to further explore non-Mendelian factors, and to improve both sequencing coverage and analytical methods. 1, 4, 5 There are many genes associated with Parkinson’s (including alpha-synuclein, which causes an autosomal dominant form of the disease) that are inherited in a Mendelian manner, however, a truly Mendelian form of Parkinson’s is relatively rare due to the interaction of Parkinson’s disease factors, which illustrates the need for clarifying the complex genetics to develop more effective treatments. 6 With more comprehensive diagnostic testing, sophisticated sequencing technologies, and more robust statistical approaches, we will one day achieve a greater understanding of non-Mendelian elements to achieve more personalized treatments to these diseases.
References
- Nagel RL (2005) “Epistasis and the genetics of human diseases.” Comptes Rendus Biologies 328(7):606-615.
- Mackay TFC & Moore JH (2014) “Why epistasis is important for tackling complex human disease genetics.” Genome Medicine 6:42 (Epub).
- Nikopoulos et al. (2019) “A frequent variant in the Japanese population determines quasi-Mendelian inheritance of rare retinal ciliopathy.” Nat Commun 10:2884 (Epub).
- Bis-Brewer DM, Fazal S & Zuchner S (2019) “Genetic Modifiers and Non-Mendelian Aspects of CMT.” Brain Res 1726:146459 (Epub).
- Kadlubowska M & Schrauwen I (2022) “Methods to Improve Molecular Diagnosis in Genomic Cold Cases in Pediatric Neurology.” Genes (Basel) 13(2):333 (Epub).
- Hernandez D, Reed X & Singleton AB (2016) “Genetics in Parkinson disease: Mendelian vs non-Mendelian inheritance.” J Neurochem 139:59-74.




