Anyone who is remotely interested in biology, or has perhaps scrolled through fitness websites to get in shape, has come across the word "protein". There is, however, much more to proteins than simply being a key player in maintaining active lifestyles. Proteins are ubiquitous in the cells of the body and are the driving force for key cellular processes. In order for proteins to carry out their duties, they need to be well-armed to execute their functions. This process of making the protein competent is achieved through specific post translational modifications (PTMs). The star of the PTMs is a cellular process called phosphorylation. The conventional methods adopted for quantifying phosphorylation are highly labor intensive. The development of phospho-specific antibodies has allowed for a huge sigh of relief from researchers due to their reputation of being quick, and detecting only phosphorylated forms of proteins in a complex mixture of phosphorylated and non-phosphorylated forms.
Why do researchers study phosphorylation?
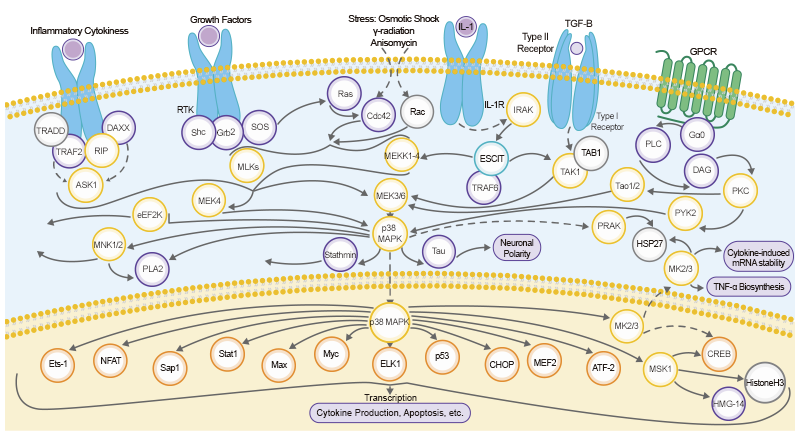
Protein phosphorylation is crucial to the regulation of a myriad of cellular processes in eukaryotes. Phosphorylation is reversible, takes place principally on serine, threonine or tyrosine residues, and is one of the most important and well-studied post-translational modifications. It plays an important physiological role in signal transduction pathways, through altering the function of transcription factors or modifying their subcellular localization.
Mechanism of Phosphorylation
The key players involved in regulation of phosphorylation are two enzymes: phosphatase and kinase. They act on their substrates, which are known as phosphate-binding proteins. Upon activation of phosphorylation by stimuli such as a tumor micro-environment, epigenetic modifications occur, including kinase's catalyzation of the transfer of the PO43- group from ATP (adenosine triphosphate) to the target protein. This is followed by dephosphorylation, which is the removal of the PO43- group by a cognate enzyme, phosphatase.
Phospho-Specific Antibodies
It has been over 30 years since the first phospho-specific antibody was produced. This first phospho-antibody, that has since been well-documented, was initially raised in rabbits against benzoyl phosphonate conjugated to keyhole limpet hemocyanin (KLH). Phosphorylation state-specific antibodies (PSSAs) have enabled the study of reversible phosphorylation of proteins that regulate nearly all key events of cell life. PSSAs have also helped researchers in their visualization of phosphorylation both in vitro and in vivo.
Phospho-Specific Antibodies in Action
PSSAs have a wide scope spanning various areas of biological research, including but not limited to: Epigenetics & Nuclear Signaling, Signal Transduction, Cell Biology & Developmental Biology, Endocrine & Metabolism, and Immunology & Inflammation. The ever-increasing demand for phospho-specific antibodies in research today can be observed through increased sales of our phospho-specific antibodies in the past year as compared to other modifications such as acetylation and methylation.
PSSAs also have potential to be widely-employed in the field of oncology, specifically in evaluating the efficacy of therapies targeting protein kinase cascades. This area of application seems especially promising, as the scope for candidate small molecule inhibitors and others in the pharmaceutical pipeline is burgeoning by the day. Evidence backed by research was seen in a xenograft model of human pancreatic adenocarcinoma. Quantitative immunofluorescence analysis of the phosphorylation state of EGFR, ERK, and PKB for the evaluation of efficacy of epidermal growth factor receptor (EGFR) inhibitor alone and in combination with wortmannin yielded useful results about the ratio of phosphorylated to total kinase protein in the same section of the viable tumor. This method, using immunofluorescence microscopy, was in agreement with the pathway blockade assessed by the administered drugs.
ABclonal offers a variety of phosphorylated antibodies covering popular research areas including epigenetics, DNA damage/repair, cancer, and immunology. Over 500 phosphorylated antibodies with multiple applications are currently available in our inventory. ABclonal is also committed to continued development of additional phosphorylated antibodies to meet the growing market demand.
Additionally, if you're curious about the various applications of antibodies in research, check out some of our other antibody-related blogs that we've curated here.
References:
- Stoevesandt, O., & Taussig, M. J. (2013). Phospho-specific antibodies by design. Nature biotechnology, 31(10), 889-891.
- Hunter, T. (1995). Protein kinases and phosphatases: the yin and yang of protein phosphorylation and signaling. Cell, 80(2), 225-236.
- Mandell, James W. “Phosphorylation state-specific antibodies: applications in investigative and diagnostic pathology.” The American journal of pathology 163,5 (2003): 1687-98. doi:10.1016/S0002-9440(10)63525-0
- Ardito, F., Giuliani, M., Perrone, D., Troiano, G., & Lo Muzio, L. (2017). The crucial role of protein phosphorylation in cell signaling and its use as targeted therapy (Review). International journal of molecular medicine, 40(2), 271–280. https://doi.org/10.3892/ijmm.2017.3036