It’s that time of the year again where we’re supposed to gather with family and close friends, talk about anything but politics, and eat monstrous amounts of good food throughout the next few days as we celebrate Thanksgiving. I used to joke with my friends that we would all be subjected to tryptophan poisoning, but just as the story about Ben Franklin wanting the national bird to be a turkey is just a myth, we can’t blame our post-feast stupor on just tryptophan either.
It’s that time of the year again where we’re supposed to gather with family and close friends, talk about anything but politics, and eat monstrous amounts of good food throughout the next few days as we celebrate Thanksgiving. I used to joke with my friends that we would all be subjected to tryptophan poisoning, but just as the story about Ben Franklin wanting the national bird to be a turkey is just a myth, we can’t blame our post-feast stupor on just tryptophan either.
Let’s Get the Myth Out of the Way…
 While tryptophan is an essential amino acid that humans must get from their diet, and turkeys happen to be a good source of it, tryptophan alone is not the reason that we crash after our bountiful feasting. It is true that tryptophan gives rise to metabolites such as serotonin and melatonin, (Figure 1)1, 2 which controls our sleep cycle, but there simply isn’t enough tryptophan in the turkey you eat (unless you’re Joey Chestnut) to knock you out. It appears that the culprit is mostly the carbohydrates you eat, in addition to the turkey, that leads to sleepiness. Think of the cranberry sauce, mashed potatoes, corn, macaroni salad on the side, and the pie(s) and you can see why there might be a sugar crash after your meal. And in late November, when the Earth is about to enter the winter solstice, especially if you are in more northern latitudes, the sun will set sooner and that will obviously affect your circadian rhythm.
While tryptophan is an essential amino acid that humans must get from their diet, and turkeys happen to be a good source of it, tryptophan alone is not the reason that we crash after our bountiful feasting. It is true that tryptophan gives rise to metabolites such as serotonin and melatonin, (Figure 1)1, 2 which controls our sleep cycle, but there simply isn’t enough tryptophan in the turkey you eat (unless you’re Joey Chestnut) to knock you out. It appears that the culprit is mostly the carbohydrates you eat, in addition to the turkey, that leads to sleepiness. Think of the cranberry sauce, mashed potatoes, corn, macaroni salad on the side, and the pie(s) and you can see why there might be a sugar crash after your meal. And in late November, when the Earth is about to enter the winter solstice, especially if you are in more northern latitudes, the sun will set sooner and that will obviously affect your circadian rhythm.
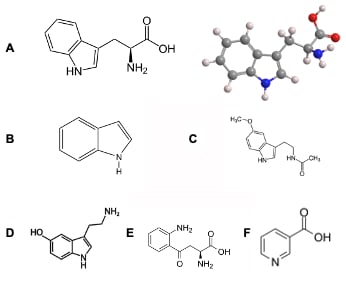
Figure 1 (above right): Tryptophan and its metabolites. A) The structure of tryptophan (left) and a molecular model of the amino acid (right). B) The indole moiety that forms the side chain of tryptophan. Derivatives of tryptophan such as C) melatonin and D) serotonin affect mood and sleep-wake cycles. E) Kynurenine is the primary metabolite of tryptophan and can be further converted into important vitamins such as F) niacin, or Vitamin B3.
Tryptophan is Quite Useful
Humans need to keep eating the right foods to ensure the replenishment of essential amino acids like tryptophan that cannot be synthesized by the body. Once in the body, the tryptophan can either be broken down into downstream metabolites (Figure 1) or loaded onto its cognate transfer RNA through the action of tryptophanyl-tRNA synthetase 1 to be incorporated into newly synthesized proteins whenever the ribosome sees a UGG codon 2 (which is just one step away from a UGA, and we certainly don’t want the protein to be prematurely truncated!).
 As the largest amino acid, tryptophan is also the scarcest, requiring much energy to process in the body while giving rise to a plethora of essential biological molecules in key functions. Niacin, for example, is an end product of the kynurenine pathway, and is important for the formation of nicotinamide adenine dinucleotide (NAD) and its phosphate (NADP), 2 which are in turn important for various metabolic functions including in cellular respiration and biosynthetic pathways. We already discussed the turkey sleep myth above, but tryptophan metabolites do control our sleep-wake cycles through melatonin and serotonin. 1, 2
As the largest amino acid, tryptophan is also the scarcest, requiring much energy to process in the body while giving rise to a plethora of essential biological molecules in key functions. Niacin, for example, is an end product of the kynurenine pathway, and is important for the formation of nicotinamide adenine dinucleotide (NAD) and its phosphate (NADP), 2 which are in turn important for various metabolic functions including in cellular respiration and biosynthetic pathways. We already discussed the turkey sleep myth above, but tryptophan metabolites do control our sleep-wake cycles through melatonin and serotonin. 1, 2
The molecules derived from indole include indolic acids, which have antioxidant properties. 1 The tryptophan is normally directed to the kynurenine pathway, but a small percentage of tryptophan is used to generate serotonin. Kynurenine and serotonin act in balance to regulate immune cell function and the inflammatory response, and alterations in this balance are associated with gastrointestinal diseases such as inflammatory bowel disease. 1, 3
Tryptophan in Health and Disease
As with many phenomena in nature, a careful balance is required to maintain optimal health in organisms, including humans. Maintaining a healthy level of tryptophan is key to generating the critical metabolites the body needs for many functions, but too much will lead to the generation of toxic compounds that could lead to inflammation and degeneration. 1-3
I was intrigued to read that activated T cells can lead to tryptophan shortages, which I initially thought would just arrest translation whenever a ribosome runs across the UGG codon. However, a recent publication showed that protein synthesis would continue in-frame across UGG codons even if tryptophan is scarce, simply replacing the missing amino acid with a phenylalanine. 4 This compensatory amino acid substitution would free up an otherwise clogged ribosome but is also reported to be associated with multiple cancer types. 4
 Tangentially, because tryptophan leads to kynurenine and kynurenine metabolites can influence inflammation, not only in the gut but also in other parts of the body including the brain, this might affect one’s mood. A recent study of depression in both pregnant and postpartum women measured the levels of proinflammatory cytokines and kynurenine metabolites in these women. They found positive correlation between IL-1β and IL-6 levels and more severe depression, and a combination of these cytokines and kynurenine metabolites had a greater than 99% probability of predicting depression. 5
Tangentially, because tryptophan leads to kynurenine and kynurenine metabolites can influence inflammation, not only in the gut but also in other parts of the body including the brain, this might affect one’s mood. A recent study of depression in both pregnant and postpartum women measured the levels of proinflammatory cytokines and kynurenine metabolites in these women. They found positive correlation between IL-1β and IL-6 levels and more severe depression, and a combination of these cytokines and kynurenine metabolites had a greater than 99% probability of predicting depression. 5
Be Thankful for Tryptophan
While I knew tryptophan to be essential to life, I never really appreciated how much a single amino acid could do to maintain healthy existence. Research has shown that the proper dosage of tryptophan through diet is key to good health, but also shows the problems that arise from excess or disruption of metabolic pathways involving tryptophan. 1 Future research will focus on how to modulate tryptophan and its pathways to avert aberrant inflammation and toxicity while boosting our quality of life. Because targeted approaches are all the rage now, most likely many scientists are working to innovate therapies directed toward the various enzymes in the tryptophan pathways.
And in the meantime, enjoy your turkey day with your loved ones! The amount of tryptophan you take in most likely won’t kill you.
References
- Poeggeler B, Singh SK, and Pappolla MA (2022) “Tryptophan in Nutrition and Health.” Int J Mol Sci 23(10):5455 (Epub).
- Barik S (2020) “The Uniqueness of Tryptophan in Biology: Properties, Metabolism, Interactions, and Localization in Proteins.” Int J Mol Sci 21(22):8776 (Epub).
- Haq S, Grondin JA, and Khan WI (2021) “Tryptophan-derived serotonin-kynurenine balance in immune activation and intestinal inflammation.” FASEB J 35(10):e21888 (Epub).
- Pataskar et al. (2022) “Tryptophan depletion results in tryptophan-to-phenylalanine substituants.” Nature 603(7902):721-727.
- Sha et al. (2022) “Cytokines and tryptophan metabolites can predict depressive symptoms in pregnancy.” Transl Psychiatry 12:35 (Epub).