In March 2022, the United States Senate approved the Sunshine Protection Act, which would make Daylight Savings Time (DST) permanent starting in November of 2023. There was still some healthy debate over whether Americans should accept Standard Time versus DST as their new permanent or keep the current system of “spring forward, fall back.” Regardless of whether we will have DST forever, there is broad consensus that the clock switch every March and November is disruptive to our sleep patterns and our circadian rhythms.
Whether to save energy, increase night-time Trick-or-Treat hours on Halloween, get those few extra minutes of sun to squeeze in the last innings of a Little League or high school baseball game, or just to normalize our sleep patterns, even a seemingly obscure issue like switching between standard time and DST is tied to our health and well-being in our society. And this is why we have to consider how sleep and the circadian rhythm can affect our physiology.
Day After Day: 24 Hours of Circadian Rhythm
As life evolved on planet Earth over the past 3.5 billion years or so, the length of a single day due to the Earth’s rotation didn’t change much. The daily duration isn’t exactly 24 hours (which is why we have leap seconds and leap years to synchronize our calendars), as it fluctuates depending on how fast Earth decides to spin on any given day. And apparently, the Moon is very gradually slowing the Earth’s rotation due to tidal friction, which adds a couple milliseconds to the day about every century. Just like how plants and other photosynthetic organisms evolved as if saying, “Hey, the sun is here and has always been here, might as well use it to make food” (at least until the theoretical expansion into a red giant that swallows the inner planets, but we won’t be able to do much about that, and it’s not going to happen anyway for a few billion years), organisms on Earth evolved to use the nearly perfectly consistent 24 hour clock to govern their natural rhythm. (Figure 1)

Figure 1: Since Earth's day has been about 24 hours long since time immemorial, all organisms logically evolved a biological rhythm corresponding to this clock.
It has always been apparent that living things, from plants and mushroom to animals, display physiology that oscillates depending on the time of day. However, scientists didn’t know how this biological clock was maintained. The mechanism of circadian rhythm began to be dissected when Jeffrey Hall, Michael Rosbash, and Michael Young isolated the period gene in 1984, which led to further discoveries that culminated in a better understanding of our biological clock and was deserving of the 2017 Nobel Prize in Physiology or Medicine. Not only did this landmark discovery help us understand the biochemistry behind the biological clock, but it also allowed doctors, nutritionists, and others to develop better lifestyle strategies to leverage the circadian rhythm in promoting optimal health. For an overview of how to optimize your circadian rhythm for your lifestyle, check out this TED Talk from leading circadian biologist Satchin Panda.
What Makes You Tick: Mechanisms Behind the Circadian Rhythm
As the world becomes more digitized and everyone carries around a smartphone nowadays, screen time will increase commensurately due to work and our own obsessive “doom scrolling” on Twitter, so that also means a lot more artificial light coming our way in the future. You may have noticed that many optometrists are now recommending blue light filters if you wear corrective lenses, and there are now commercially available blue light filters you can use to shield your monitor. They’re even available on your handheld devices! This is because, even though blue only comprises one part of the spectrum, the blue light from natural sunlight and from your screens can disrupt your circadian rhythm, and can contribute to disease states. 1, 2
Environmental light input is received by specialized cells in the retina and then transmitted to the suprachiasmatic nucleus (SCN), a part of the brain next to the hypothalamus. 3 The SCN serves as a key component of the circadian clock, employing tightly regulated nervous and hormonal activities to control multiple physiological functions throughout the 24-hour cycle. 4 One of the chief functions of the SCN is regulating melatonin production by the pineal gland, which also feeds back on the SCN in a coordinated fashion, and this contributes to habitual sleep patterns in mammals including humans. 5, 6 (Figure 2) Because light is the critical input that suppresses melatonin production and has the ability to reset the circadian cycle, this mechanism is heavily studied in sleep disorders and jet lag. 3, 7, 8
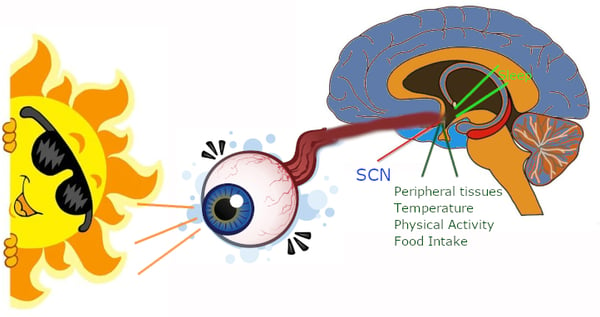
Figure 2: Light stimuli via the retina is relayed to the suprachiasmatic nucleus (SCN), which regulates the circadian function of body systems.
The elegant molecular machinery that drives the circadian rhythm differs between organisms but has the shared evolutionary function of being oscillators that follow the 24-hour rotation of the Earth. In most eukaryotes, including mammals, the circadian oscillator is a transcription-translation feedback loop, where the components of the loop alternate waves of activation and repression. This oscillation is aided in part by the short half-life of both the transcripts and the protein products, and a typical feedback mechanism comprises the classical circadian proteins CLOCK, BMAL1, PERIOD, Cryptochrome, ROR, and REV-ERB. (Figure 3) 2, 9 The intricate management of the circadian rhythm allows organisms to be active and rest at the proper times of day, ensuring that their systems are operating at peak efficiency during the appropriate parts of the cycle.
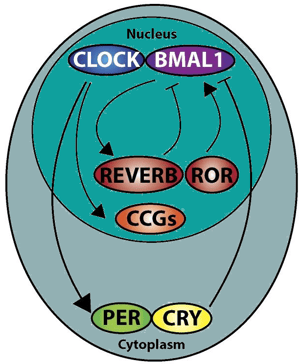
Figure 3: The transcription-translation feedback loop in mammalian circadian oscillation. Adapted from Chaix et al. (2016) 9
Shining a Light On the Circadian Rhythm In Maintaining Health
A functional circadian clock is key to the proper cycling of organismal behavior and physiology, which include the sleep-wake cycles but also the optimal function of body systems, metabolism, immunity, and more. 2, 3, 7, 8, 9 Looking deeper into individual cells, circadian regulation is key to the timing of transcription and posttranslational modifications, organelle function, and the cell cycle. 9 Disruption to circadian rhythms are correlated with a number of physiological issues, including diabetes, obesity, and jet lag as mentioned above. 2, 7 Due to its role in maintaining properly cycling of metabolic states, circadian dysregulation has been linked to cancer and immune disorders. 1, 2
Circadian control over most body functions can be leveraged to not only provide blue light filters and reduce jet lag, but also to determine the best times of the day to take medications. In a recent study using ABclonal antibodies, Quattrocelli et al. reported that the effect of prednisone on muscle bioenergetics required a functional circadian clock. (Figure 4) 10 Administering the medication at the start of the light cycle significantly improved its effects, and led to increased mitochondrial density, an indicator of improved muscle bioenergetics. 10
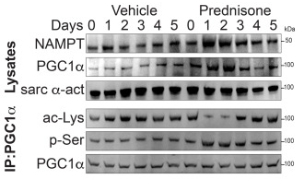
Figure 4: Prednisone treatment compared with control (vehicle) at ZT0 of the light cycle. The peroxisome proliferator-activated receptor-γ coactivator 1α (PGC1α) shows an activation profile with decreased acetylation and increased phosphorylation. The nicotinamide phosphoribosyltransferase (NAMPT) also increases in expression over the short term, signifying increased metabolic rate. Source: Quattrocelli et al. (2022) 10
Together, the discoveries and epiphanies regarding the circadian rhythm, ever since the landmark discoveries of Hall, Rosbash, and Young, allow us to live better, healthier lives through coordination of our activities according to the natural light-dark cycles that the planet has experienced since the formation of our solar system. Through both biochemical therapy and behavior modification, we can improve our quality of life and reduce the prevalence of diseases. At the minimum, the biology is very clear on the benefits of a good night’s sleep, which we could all use!
How ABclonal Can Help
As seen in the report by Quattrocelli et al., ABclonal Technology provides a plethora of antibody reagents and ELISA kits suitable for the study of the circadian rhythm. 10 A sample of the ABclonal reagents used in their study is listed below, and we hope that we can assist you in uncovering new insights into leveraging the circadian rhythm to promote global health.
|
Cat. No. |
Product |
|
Glucocorticoid Receptor Rabbit pAb |
|
|
BMAL1 Rabbit mAb |
|
|
PBEF / NAMPT Rabbit pAb |
|
|
PGC1α Rabbit pAb |
|
|
Rev-Erbα / NR1D1 Rabbit mAb |
References
- Zhang et al. (2021) “Circadian clock: a regulator of the immunity in cancer.” Cell Communication and Signaling 19:37.
- Patke et al. (2020) “Molecular mechanisms and physiological importance of circadian rhythms.” Nature Reviews Molecular Cell Biology 21:67-84.
- Blume et al. (2019) “Effects of light on human circadian rhythms, sleep and mood.” Somnologie (Berl) 23(3):147-156.
- Hastings et al. (2018) “Generation of circadian rhythms in the suprachiasmatic nucleus.” Nature Reviews Neuroscience 19:453-469.
- Gillette and McArthur (1996) “Circadian actions of melatonin at the suprachiasmatic nucleus.” Behav Brain Res 73(1-2):135-139.
- Brennan et al. (2007) “Light, dark, and melatonin: emerging evidence for the importance of melatonin in ocular physiology.” Eye 21:901-908.
- Vosko et al. (2010) “Jet lag syndrome: circadian organization, pathophysiology, and management strategies.” Nature and Science of Sleep 2:187-198.
- Stenvers et al. (2016) “Dim light at night disturbs the daily sleep-wake cycle in the rat.” Scientific Reports 6:35662.
- Chaix et al. (2016) “The circadian coordination of cell biology.” J Cell Biol 215(1):15-25
- Quattrocelli et al. (2022) “Muscle mitochondrial remodeling by intermittent glucocorticoid drugs requires an intact circadian clock and muscle PGC1a.” Sci Adv 8(7):eabm1189 (Epub).




