Optimizing IF Outcomes Through Rational Antibody Selection
ABclonal x YCharOS: Setting the Gold Standard in Antibody Characterization
Detailed Guideline on Pyroptosis Research
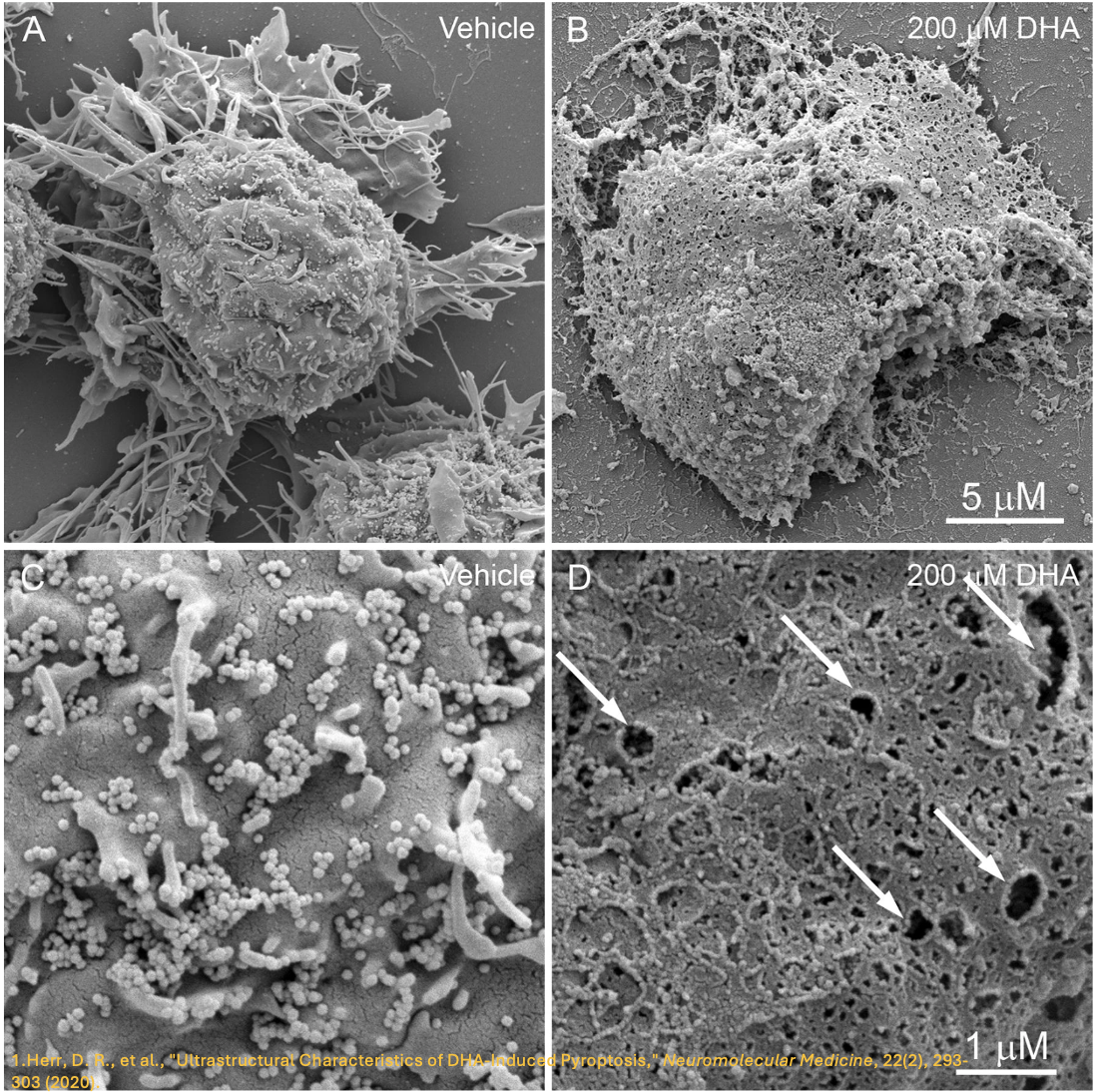
It is evident that the gasdermin family is central to pyroptosis. This is because the gasdermin family members are the executioners of pyroptosis, and their pore-forming function is a necessary condition for pyroptosis to occur. This family generally includes a cytotoxic N-terminal domain and a C-terminal inhibitory domain. After cleavage, the N-terminal domain is released and can assemble into pores in the membrane. Gasdermin pores disrupt the integrity of the cell membrane, leading to inflammatory cell death, with cell contents, including inflammatory cytokines, being released into the extracellular space.
In the 1980s and 1990s, scientists discovered that macrophages exposed to bacterial toxins or infections underwent a unique form of cell death that required the activation of caspase-1. For years, this process was mistakenly classified as apoptosis, a non-inflammatory and orderly form of cell death. However, unlike apoptosis, this newly observed cell death was marked by swelling, rupture, and the release of inflammatory signals, indicating a far more chaotic and immune-activating process. It wasn’t until 2001 that Cookson BT and Brennan MA officially named this inflammatory form of cell death “pyroptosis”. [2] Since then, research has revealed that pyroptosis plays a key role in immune defense by promoting inflammation in response to infections. The discovery of gasdermin D (GSDMD), a protein that forms pores in the cell membrane, further clarified the mechanism of pyroptosis. Today, it’s understood that pyroptosis occurs in various cell types and is critical in both fighting infections and contributing to inflammatory diseases when dysregulated.
Although the phenomenon of cell death had been known for centuries, even briefly described in the 19th century, the explosion of research and technology in the latter half of the 20th century led to greater and more nuanced discoveries that have provided insights into many physiological processes including tissue development, maintenance, metabolism, and disease states including cancer. The many accomplishments in programmed cell death research have improved our understanding and development of targeted therapeutics, and some of these milestones were recognized by Nobel Prizes for apoptosis and autophagy. As scientists continue to elucidate new cell death pathways and their interplay with other pathways, let's take a look at what we know so far and what new findings have come out.