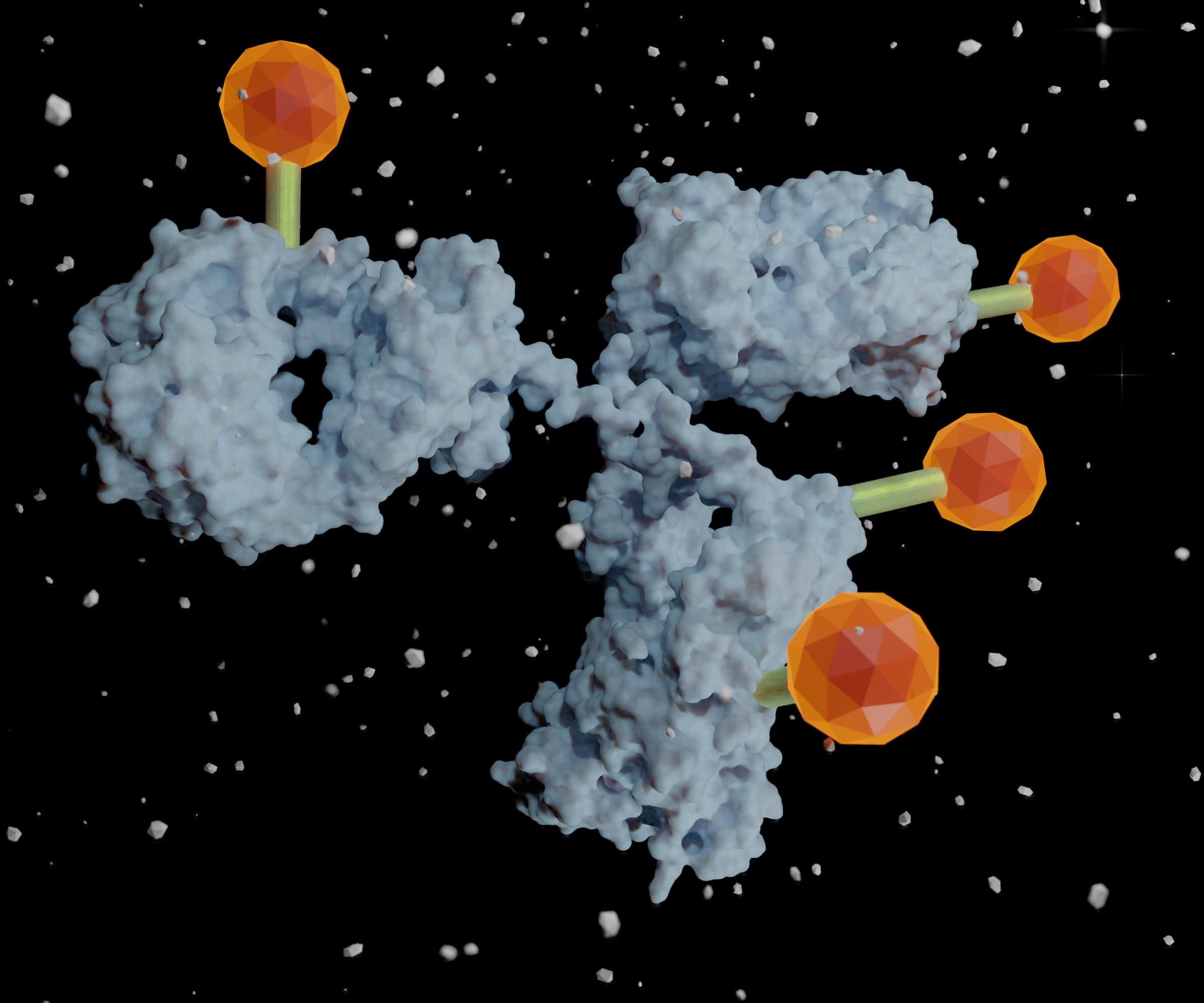
The dream of biomedical researchers is to fine-tune their therapeutics to precisely target the specific illness or pathogen affecting their patient. Ever since Nobel laureate and oft-quoted father of immunology, Paul Ehrlich, coined the term “magic bullet,” medical science has marched towards more personalized drugs that target key molecules that cause diseases including cancer. 1 We find ourselves now, over a century later, in an exciting era of discovery that has produced many antibody drug conjugates (ADC) designed to precisely target the diseased cells and not healthy cells. ADC uses this strategy to take advantage of the specificity of antibodies while delivering a covalently linked cytotoxic payload directly to diseased tissues to reduce the multitudes of side effects and toxicity. 2, 3 As basic research identifies more targets and antibody engineering procedures improve, the range of antitumor and anti-disease weapons may seem limitless.
The Magic Bullet: Current State of the Antibody-Drug Conjugate Market
As science advances, one of the recent trends that continues to pay dividends is immunotherapy to fight cancers. In many cases, the strategy is to mobilize the immune system to attack tumor cells based on cancer-specific antigens expressed either by the tumor itself or within the tumor microenvironment, either by stimulating normal immune cell function to their new tumor target or by removing the suppression of the immune system that is characteristic of many cancers. The trick is to find a way to attack only the tumor and not normal tissues, and certainly not to somehow trigger autoimmunity.
Computers are ubiquitous in our lives now, particularly the majority of us who have a miniature supercomputer in the palm of our hands. With more efficient and powerful computer technology coupled with the understanding that biology is much more complex than dissecting out the role of a single protein in a signaling pathway, the relatively new field of computational biology and bioinformatics has exploded over the past few decades as researchers needed new tools and strategies to understand biology on a systems level. This has allowed non-traditional professionals to enter bioscience research, from primarily computer scientists to bench scientists who have taught themselves coding and statistics. As the computational component has permeated through nearly all of modern biology, we realize that there is a beneficial coexistence between the experimentalists and the keyboard warriors who make sense of growing datasets.
Remember once upon a time when I said my first actual laboratory research project involved myelin basic protein? Other than knowing that the mother of one of my high school friends had been diagnosed with it, this was the first real exposure I had with multiple sclerosis. I eventually learned more about the immune system and autoimmunity, and the thought of your own body attacking your literal nerve cells was scary and made me feel for the people who have to live with and manage this disease every day. March happens to be Multiple Sclerosis Awareness Month, which as the name suggests works to make sure the public knows about multiple sclerosis, develops empathy and understanding for afflicted individuals, and encourages participation in events and activities to spread awareness. In this blog, let's explore the disease, current treatment strategies and ongoing research, and ways that you can help both in and out of the lab.
What is STAT5B?
STAT5B belongs to the STAT (signal transducer and activator of transcription) protein family, a group of latent cytosolic transcription factors activated by Janus kinase (JAK) tyrosine kinases. The JAK-STAT signaling pathway is responsible for many important biological processes including cell proliferation, differentiation, apoptosis, and is also involved in the modulation of a variety of cytokines to control the immune response.
Trim and Proper: A Nifty Method for Targeted Protein Degradation
A common experimental strategy in studying the effects of a specific protein in cells or organisms is to remove it. One can determine the physiological outcomes in the absence of that protein to ascertain its relative importance in maintaining normal functions, or in some cases, to note that it is dispensable or redundant and might have a backup within the cell to take up the slack. Some targeted techniques include RNA interference (RNAi) and CRISPR-based gene editing, and in many cases, it is possible to generate knockout cell lines or even organisms, like mice, that cannot express a specific protein. But when those strategies are not feasible for the experiment at hand, what is one to do?